Tailoring electrolyte solvation for Li metal batteries cycled at ultra-low temperature. Self-smoothing anode for achieving high-energy lithium metal batteries under realistic conditions. Our work provides insights into the interplay between the molecular structure of Li salts, their physicochemical properties and electrochemical performances. Under fast-cycling conditions (charging: 1.46 mA cm − 2, discharging: 3.66 mA cm − 2), pouch cells maintained 81% capacity after 100 cycles. Pouch cells of 310 Wh kg −1 achieved ~410 W kg −1 power density at the discharging current density of 6.59 mA cm −2. 
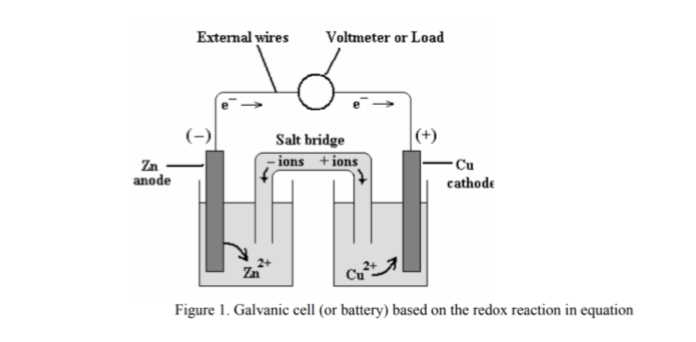
LiFEA-based carbonate electrolytes notably improved fast-cycling performances of Li | |NCM811 cells. It enables carbonate electrolytes with a large apparent donor number and Li + transference number and drives a self-cleaning mechanism for solid–electrolyte interphases, enhancing compatibility with Li-metal anodes even at high current densities. Herein we design an asymmetric Li salt, lithium 1,1,1-trifluoro- N-ethyl] methanesulfonamide (LiFEA) that possesses a pseudo-crown ether-like, folded molecular geometry. In the reaction, the silver ion is reduced by gaining an electron, and solid Ag is the cathode.Conventional carbonate-based electrolytes with high corrosion towards Li metal result in massive dendrite growth and limited cycling life, particularly true for practical Li-metal batteries with high cathode loading (>3.5 mAh cm − 2). In a typical voltaic cell, the redox pair is copper and zinc, represented in the following half-cell reactions: This redox reaction consists of two half-reactions. The operating principle of the voltaic cell is a simultaneous oxidation and reduction reaction, called a redox reaction. This flow of electrons is an electrical current that can be used to do work, such as turn a motor or power a light. The two electrodes must be electrically connected to each other, allowing for a flow of electrons that leave the metal of the anode and flow through this connection to the ions at the surface of the cathode. This forms a solid metal that deposits on the cathode. At the cathode, the metal ion in the solution will accept one or more electrons from the cathode, and the ion’s oxidation state will reduce to 0. The metal of the anode will oxidize, going from an oxidation state of 0 (in the solid form) to a positive oxidation state, and it will become an ion. The anode will undergo oxidation and the cathode will undergo reduction. The voltaic cell uses two different metal electrodes, each in an electrolyte solution. In between these electrodes is the electrolyte, which contains ions that can freely move. 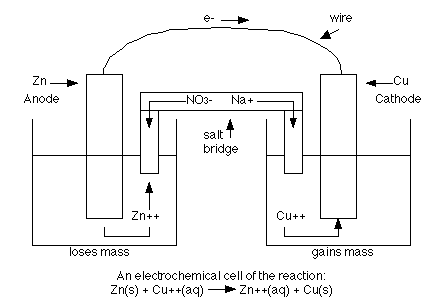
Electrodes can be made from any sufficiently conductive materials, such as metals, semiconductors, graphite, and even conductive polymers. The cathode is the electrode where reduction takes place. The anode is defined as the electrode where oxidation occurs. These scientists conducted several experiments on chemical reactions and electric current during the late 18th century.Įlectrochemical cells have two conductive electrodes, called the anode and the cathode. This kind of cell includes the galvanic, or voltaic, cell, named after Luigi Galvani and Alessandro Volta.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
